It was a hot July 1928, and Scottish scientist Dr. Alexander Fleming had long been fascinated by the harmful effects of bacteria and other microorganisms. After working as a researcher at St. Mary’s Hospital in London, England, for almost fifteen years, Dr. Fleming had taken a vacation with his wife and young son to his country house in Suffolk. Looking after his petri dishes containing growing strains of the bacterial species Staphylococcus aureus was his research assistant Dr. Merlin Pryce. After a month of vacation with his family, Dr. Fleming returned to the lab with Pryce.1 Both had foretold that the bacteria strain had grown in all petri dishes; but neither scientist could have foretold what grew with it.

Fleming and Pryce had started to discard the petri dishes, but as Fleming handed a random dish to his assistant, both noticed something quite peculiar: a bluish-green mold had grown with the culture of staph. This growth was not an uncommon characteristic of bacterial growth in petri dishes, and Fleming himself explained that the growth of other species reflects the laboratory technique of the scientist.2 Upon closer inspection, however, both scientists found something even more strange: no bacteria grew around this mold for a considerable distance. Fleming had initially believed that the substance produced by the mold was similar to a lysozyme, an enzyme found in human tears that kills germs.3 Both scientists concluded that something produced from this mysterious mold had caused the microorganisms to die, a substance that, at the time, quickly became the most powerful drug known to medicine: penicillin.
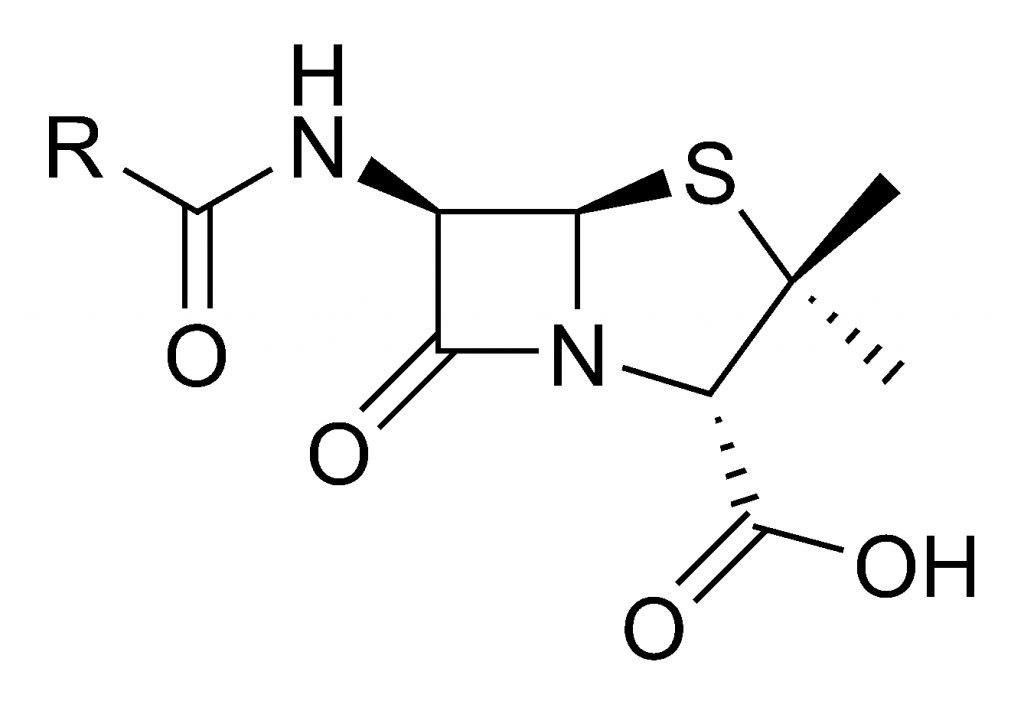
All bacteria are prokaryotic in nature; these organisms are free-living cells with no nucleus to encompass genetic information. All bacteria contain circular DNA and ribosomes used to create proteins with many different functions used throughout different parts of these cells. Despite these similarities, “bacteria” describes a superkingdom of organisms with a wide range of diversity. These incredibly small unicellular bodies have a bad reputation for causing disease, but in reality, only about one in one thousand species of bacteria are actually harmful to humans.4 Depending on the genetic information provided and the proteins produced, some species are not only beneficial, but necessary, for our survival; some strains are able digest food, some ensure that blood flows properly, and others undergo photosynthesis to release oxygen into the atmosphere. Strains that are harmful, however, have genetic code that has been altered to become not only antibiotic-resistant, but detrimental to our health. Ailments like tetanus, tuberculosis, acne, and even sexually transmitted infections like chlamydia and syphilis are all caused by different strains of bacteria with different genetic information corresponding to each disease.5 Staphylococcus aureus, more commonly known as the “staph infection,” was no different from every other pathogen in the sense of causing pain and suffering to those without an effective form of treatment. Although readily treated today, diseases like the staph infection had claimed the lives of countless people not only in Europe, but all over the world up to the early twentieth century; however, these pathogens would eventually meet their match. Penicillin is a molecule that inhibits the formation of binding proteins in bacterial cell walls. Once these proteins can no longer bring parts of the cell wall together, bacterial cells disintegrate and die.6 After Fleming and Pryce made this startling discovery, both scientists, and Fleming’s longtime assistant Stuart Craddock, worked tirelessly to see what else this mysterious substance could do. All quickly discovered that along with strains of Staphylococcus and Streptococcus, more commonly known as “strep throat,” stable forms of this substance effectively kill bacteria that cause pneumonia, meningitis, and more. Even more exciting to discover, this substance did not harm white blood cells, and when injected into rabbits and mice, it was determined to be non-toxic. In January 1929, Dr. Craddock had suffered from a sinus infection, an ailment that Dr. Fleming turned into a striking advantage for this substance’s power. After swabbing Dr. Craddock’s nose and counting the number of bacteria present, Dr. Fleming injected one cubic centimeter of this substance into Craddock’s nostril. Within three hours, about 99 percent of the bacteria was gone.7After these successful experiments, Fleming had desired to separate the substance from the mold that grew with it. Unfortunately, Pryce and Craddock had their time invested in other research projects, and could not assist Dr. Fleming with this substance any longer. Fleming tried using various techniques for substance separation that he had used for other particles in the past, but to no avail. The mold that had grown in the petri dish was clarified as Penicillium notatum by American botanist–and Fleming’s friend–Charles Thom. So, Fleming had given the name “penicillin” to the seemingly inseparable “mold juice.”8 After more failed attempts to extract penicillin from its source, Fleming had abandoned the project, concluding that although effective, penicillin is too unstable, breaks down too easily, and is very difficult to produce.9 In 1929, Fleming had published his findings in the British Journal of Experimental Pathology, an article that pathologist Howard Florey had read and had drawn inspiration.

Dr. Florey, the Australian-born head of the William Dunn School of Pathology at the University of Oxford, was deeply interested in the discovery of penicillin. As a scientist with an inclination towards chemistry, he wanted the subject to be more incorporated in the study of pathology, and believed that expanding on Dr. Fleming’s findings would prove to become the perfect example of this incorporation. In 1937, nearly a decade after Dr. Fleming’s discovery, Florey had recruited the smart and determined biochemist Ernst Chain immediately after Chain finished his second doctorate at the University of Cambridge. As a Jewish refugee from Nazi Germany, Chain was grateful and ready for the opportunity to pursue his passion for research during times of prejudice and heartbreaking cruelty.10
By this time, penicillin was described as “[a molecule] as unstable as an opera singer,” and bacteriologists and biochemists all over London failed to separate this substance from the mold. But, Florey, Chain, and their team at Oxford used a technique called freeze-drying, a process normally used to make blood plasma for the war effort. Although this molecule can now be separated, the samples of penicillin used by Florey and Chain’s team were very low in concentration. At approximately one part per million of the original solution, these researchers feared that not enough of this molecule could be salvaged.11 However, one prominent biochemist, Dr. Norman Heatley, had offered a striking solution.
In 1940, Dr. Heatley had proposed a way to separate penicillin from the original solution. He explained that molecules of penicillin could be transferred from the original broth to a solution of ether, a non-polar substance that does not mix with water. These molecules would then be transferred back to an alkaline solution in water, and the process would be repeated until higher amounts of pure penicillin would be left behind.12 Using this method, Dr. Heatley had built a countercurrent machine made specifically for this separation and extraction of penicillin. This contraption was made mostly out of flattened glass and copper coils, cheap materials that proved Dr. Heatley’s resourcefulness later compared to that of scientists Ernest Rutherford, the father of nuclear physics, and Otto Hahn, the man who split the atom.13 Heatley’s laboratory capabilities did not go unnoticed by Florey, who commended the biochemist, or Chain, who resented him out of jealousy. As time passed, Chain and Heatley’s relationship became so strained that Chain would only report his findings to Florey, not to Heatley. But in the context of war, catastrophe made the scientists’ work disputes seem miniscule.
Approximately 60 million people had perished during the course of World War II. With the rise of anti-Semitism and cruelty in the Nazi Party, 9 to 11 million of these people have lost their lives in concentration camps alone. The attack on Pearl Harbor, the bombings of Hiroshima and Nagasaki, the Battle of Normandy, and the mass genocide of 6 million Jews in Auschwitz, Bergen-Belsen, and other death camps prove to exemplify the true horrors of war.14 The amount of those afflicted by typhus and malaria were innumerable, with countless soldiers perishing in battle not just from gunshot wounds, but from disease. The British Eighth Army in Italy, for example, was no exception in terms of contracting these diseases; at this time, approximately 41 percent of troops per year were either confirmed or suspected to have contracted malaria before preventative measures were taken. A typhus epidemic had plagued Naples after malaria had ravaged Italian civilians and British soldiers alike, claiming the lives of over one thousand people before the disease was contained.15 Those living in inhumane conditions were in desperate need of support, and mass production proved to be salvation. Florey, Chain, and Heatley had worked quickly, but meticulously, to make penicillin a useful antibiotic. After more trials for separation, tests for toxicity, and evaluation for effectiveness against bacteria, Florey had placed his faith in American industry companies like Pfizer for the mass production of penicillin. Although British industry was performing rather well, Florey knew that these companies lacked overall experience and organized collaboration compared to the United States. In the second half of 1943, American companies produced beyond the four researchers’ imaginations. Over 20 billion units of penicillin were produced by United States industry in the second half of 1943; the next year, that amount increased eighty-fold. A sharp increase in demand and vast amount of production ensured that penicillin became relatively cheap, thus branching it out to more people in need of deliverance by the newly found “wonder drug.”16
For their contributions, Drs. Fleming, Chain, and Florey received the Nobel Prize in Physiology or Medicine in 1945. Dr. Fleming had received the most public recognition for the discovery of penicillin, and was awarded several honorary doctorates, prizes, and tributes from British citizens. The names of Florey, Chain, and Heatley may have not achieved the same level of popularity as Fleming, but each had gone back to work for the advancements of scientific research. In 1963, Dr. Florey was inducted as the provost of Queen’s College, and continued to work at the Dunn School of Pathology until his retirement in 1976. Dr. Chain was inducted into knighthood in 1969, and supported the formation of the State of Israel. Although not formally recognized for the Prize, a topic still debated among scientists today, Heatley also received praise from his colleagues for his work and persistence in times of crisis; he was appointed to the Order of the British Empire in 1978.17
What could only be described as one of the most groundbreaking and revolutionary scientific discoveries, penicillin continues to be the original and a powerful antibiotic that relieves all afflicted by pain and illness. Doctors Fleming, Chain, Florey, and Heatley deservedly have their names marked in our history books, and against unimaginable pressure, they have found a way to bring healing to a world of grief, sorrow, genocide, and agony. These outstandingly talented scientists have paved the way for generations of doctors, healthcare providers, and ambitious students to pursue their careers, follow their dreams, and become figures of survival and hope.
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 16-17. ↵
- H. Landsberg. “Prelude to the Discovery of Penicillin,” Isis 40, no. 3 (August 1949): 226. ↵
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 17. ↵
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 12. ↵
- World of Microbiology and Immunology, 2003, s. v. “Chronic Bacterial Disease.” ↵
- Chemistry: Foundations and Applications, 2004, s. v. “Penicillin,” by William G. Gutheil. ↵
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 27. ↵
- Robert Bud, Penicillin: Triumph and Tragedy (Oxford: Oxford University Press, 2007), 26-27. ↵
- Jon Agar, Science in the Twentieth Century and Beyond (Cambridge: Polity Press, 2012), 266. ↵
- “Howard Florey and the Development of Penicillin,” Notes and Records of the Royal Society of London 53, no. 2 (May 1999): 246. ↵
- Robert Bud, Penicillin: Triumph and Tragedy (Oxford: Oxford University Press, 2007), 27. ↵
- Robert Bud, Penicillin: Triumph and Tragedy (Oxford: Oxford University Press, 2007), 30. ↵
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 107-108. ↵
- Gale Encylcopedia of World History: War, 2008, s. v. “World War II (1939-1945).” ↵
- John D. C. Bennett. “Medical Support for the British Eighth Army in Italy During World War II,” Army History, no. 47 (1997): 22-25. ↵
- Robert Bud, Penicillin: Triumph and Tragedy (Oxford: Oxford University Press, 2007), 46-53. ↵
- Eric Lax, The Mold in Dr. Florey Coat: The Story of the Penicillin Miracle (New York: Henry Holt and Company, 2004), 253-260. ↵


16 comments
Margaret Maguire
This article was really interesting. I never knew that it took so many people to create the penicillin we know today. I think it is really neat how many discoveries were made along the way to creating drug. I thought that the measurement of one cubic centimeter was an odd unit of measurement for a liquid type medicine, which is equal to one milliliter. I was really confused by it and looked it up. I really enjoyed reading this article because I knew what penicillin was but never knew the story behind it.
Mara Martinez
Penicillin keeps so many people in this world alive. This article was super interesting on the fact that it took so many different people to really get it made. But I had no idea, I didn’t know really anything about how it was created so this article opened my eyes to that.
Olivia Tijerina
During the reading I had time to really fathom over the works of the scientist during their time in trial because they had invested in their future when what they had little because what was in front of then was dissipating. Death from both from the problem they aspired to fix and the other was a place that had sufficiently helped the disease grow to become problematic, war. When I ponder about the Holocaust I think about how they were so vulnerable and particularly that is what disease target when there are open wounds which I can agree that all people at that time had had.
Gabriella Urrutia
I think that it is interesting to learn about penicillin because it has prevented the death of millions. With the discovery of penicillin, it was able to be used as an antibiotic for many diseases and improved the quality of life. Without it, many people would probably be suffering and end up dying from diseases. It is amazing to think about the discovery of one thing was able to help the lives of millions.
Bruno Lezama
Penicillin is the one of the most important medicine for a lot of illnesses. I think that the discovery of Penicilin crucial for human beings because without Penicillin, the were less human in the world. It is not the first time that I heard about the history of the discovery of Penicillin; however, thanks to this article, now I know more details about the accidently discover of this medicine. Great Article!
Julia Patrick
Before reading this article I had never learned about the history of penicillin. It’s very interesting to see how the drug actually works. The discovery of this drug led so many other innovative projects that could not have happened without penicillin. I’m glad I read this article because I think it’s important to know about the history of something that is so vital to the well being of society.
Briley Perkins
This is a very informative and descriptive article. I know what penicillin is, but I did not know how it was formed and who founded it. It is created by the proteins in the cell walls binding together. It is weird how things are founded or created by accident that is then used later on, because that is basically what happened with penicillin.
Paul Garza
I knew penicillin was important to the world of medicines and our present science of antibiotics, however I never knew who or how it was discovered. I like that the author included factual information that allowed the reader to stay completely engaged with the scientific talk. Crazy to think that penicillin which changed how medicine worked drastically, was dissevered through a small Petri dish and some bacteria that distanced itself. I enjoyed the closing of the article that explains how important penicillin actually is to us and how many lives it saved.
Courtney Pena
It was amazing to read about how penicillin was discovered. I thought that it was interesting how Flory had faith in the American industry company for mass production of penicillin instead of having faith in British industries due to their lack of overall experience and lack of organized collaborations. It is fascinating how all of the doctors received the Nobel Prize in Physiology or Medicine in 1945 for the discovery of penicillin. Their impact in discovering penicillin is still seen today as penicillin is known as a powerful antibiotic that can be used to heal any pain and illnesses.
Jacqueline Mendez
I have learned about Dr. Flemmming in my science classes but never to this depth. This was a very interesting article as it went deep on how penicillin works. I never knew that it bonded on the protein in the cell walls I always thought it bonded to the nucleus and DNA. A very interesting article. A good story to tell.