When a person who has cancer is faced with no more options for treatment, would you recommend they join a clinical trial? What if this person had a terminal illness, such as brain cancer, would they be able to rationally make decisions about what course to take for their own health? This is a major concern that many researchers encounter when facing clinical trials, especially when it comes to cancer, which can be a terminal illness.
All patients should be aware that when facing and enduring different difficulties throughout the course of their treatments, they might reach a certain point where the only option left is to either be part of a clinical trial or to withhold treatment. However, how would this be different in the case of brain cancer patients, when their health and mental capacities begin to deteriorate? Would it then be ethical for them to still make those important decisions for their treatment? Or would it be ethical to allow the family to make those decisions?
Brain cancer is a complex and varied disease caused by a group of abnormal cell growths that develop within the brain or surrounding structures of the central nervous system (CNS).1 There are 2 types of brain cancer: primary, which starts in the brain and does not spread to other parts of the body, and secondary (or metastases), which is mainly caused by another cancer that begins in different parts of the body.1 Cancer tumors can be classified as either benign (non-cancerous) or malignant (cancerous). It is important to know that cancer itself cannot be passed down from parents to children nor the genetic changes in tumor cells. However, what can be passed down (inherited), is any genetic change in the parent’s genome that can then provide a higher probability of being at risk but only if it is present in a parent’s egg or sperm cells.1 The primary cause of cancer is due to changes or mutations in a certain gene, which can then lead to abnormal cell growth and then divide uncontrollably and destroy body tissue. Such is the case of glioblastomas.
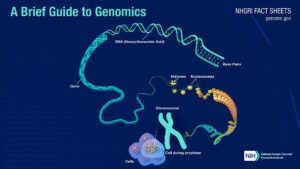
However, let’s take a step back and look at the whole picture from the start of our genetic makeup. According to the National Institute of General Medical Science, “genetics is the scientific study of genes and heredity”.2 Here certain qualities or traits can be viewed which are passed on from parents to offspring as a result of changes in DNA sequence. DNA is a chemical compound needed to develop and direct all living organisms’ activities. It is incredibly long, but tightly coiled and compacted to fit within the tiny confines of a cell’s nucleus. Genes are a segment of DNA that contains instructions for building one or more molecules that help the body work. As well as making a specific protein or set of proteins.2 Proteins are needed to help make up body structures like organs and tissue and can control chemical reactions and carry signals between cells.3All of an organism’s genetic material, including its genes and other elements that control the activity of those genes, is known as the genome. However, how does knowing this help us understand how cancer is formed? But more specifically, brain cancer.
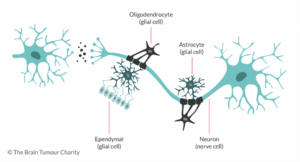
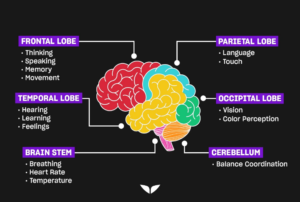
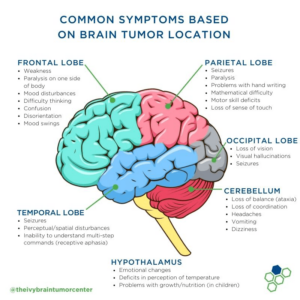
In general, when a cell’s DNA is mutated, an abnormal protein may be produced, which can disrupt the body’s usual processes and lead to a disease such as cancer.4An accumulation of genetic changes over many years can turn healthy cells into cancerous cells.5This can be greatly seen in brain cancer. For example, glioblastomas, also known as glioblastoma multiforme (GBM), are a specific type of brain cancer. GBM is considered to be one of the most aggressive and deadliest forms of cancer.6The Moffitt Cancer Center, explains that “glioblastoma is caused by DNA mutations that result in uncontrolled cell growth.” 7 It first begins in the brain’s astrocytes, which are a type of cell that provides structure and support for neurons, and they make up the majority of cells in the human central nervous system (CNS).8Glioblastoma tumors can form in any part of the brain. However, they are usually formed in the frontal lobe, which is responsible for functions like personality, emotions memory, problem-solving, impulse control, social interaction, and motor function and in the temporal lobe, which helps interpret sounds and helps recognizing and using language.9Due to this, their symptoms may vary and depend significantly on the location of the tumor. Some common symptoms seen are headaches, nausea and vomiting, changes in personality, memory, problem-solving, impulse control, social interaction, and motor function, and the temporal lobe, which helps interpret sounds and helps recognizing and using language. Therefore, some treatments to face this cancer mainly require surgery, radiation therapy, chemotherapy, and as a last resource, clinical trials.
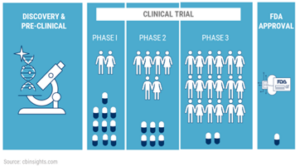
Clinical trials are an essential tool in the field of medicine and healthcare research. However, they can either be extremely helpful by obtaining promising results and data or they could lead to devastating outcomes in different cases. Nevertheless, the use of clinical trials has been extensively used to combat many illnesses. For instance, to fight the overgrowth of cells, it is key to identify the cause of that specific cancer. Once that has been obtained, they can gain a better understanding of the molecular basis of cancer growth, metastasis, and drug resistance, which is done with the use of clinical trial data. For a clinical trial to work, it must go through 4 main basic stages defined by the Food and Drug Administration (FDA), to help identify the effectiveness of the drug in different phases. In the 3 main phases, what is being established is testing the treatment, finding the correct dosages, and looking for any side effects. After those 3 first phases, if researchers are able to find a drug or other intervention to be safe and effective, it will then go to the FDA for approval for clinical use, and the FDA continues to monitor its effects. These trials are very specific research studies. Through this method, researchers can determine if a new form of treatment or prevention is safe and effective in people.10 However, going back to our initial question: is it ethical for a brain cancer patient, specifically someone with glioblastoma, to decide to participate in a clinical trial?
First, it is essential to recognize the severity and combination of symptoms among glioblastoma patients. We know that the size of the tumor, as well as the pressure it exerts on surrounding brain tissue, can grow in any part of the brain. While the specific location of it can influence the symptoms experienced by the affected individual, as well as the potential treatment options.

Many researchers who are constantly trying to find new ways to help brain cancer patients can mainly only resort to clinical trials. Unfortunately, some of those clinical trials may not always lead to immediate benefits for glioblastoma patients. Some ethical issues that can be faced in this situation are: patients are at a high risk of impaired medical decision-making capacity (MDC). 11 This can be challenging because it is limits their ability to give informed consent to any medical treatments or participation in research. Also, some of these patients may be in an emotionally vulnerable state or the tumor suppress a specific brain region in which they aren’t their normal selves. In other words, it is common for GBM patients to experience memory loss and even personality changes. Therefore, providing them with information and the option to choose to join a clinical trial after everything they had already gone through, may not be ethical.
Another issue arises due to the lack of effective treatment options. These patients’ prognosis is generally poor, and they have no more options left other than an experimental treatment. This leads to a new ethical debate: whether it is ethical to offer experimental treatments with potentially low chances of success or to provide more palliative care. This can be controversial because this decision greatly depends on whether the patient is truly willing to consent without feeling coerced or desperate for any potential experimental treatment in hopes of getting better.
In addition, many clinical trials are required to have a placebo control group, which means that there will always be one group that will get the active treatment, while the other group will just get the placebo. This is used to establish the efficacy of a new treatment. However, when facing a life-threatening disease like glioblastoma, how do you choose who will receive what? Doctors and/or researchers are the ones responsible for assigning the patients to a group that will receive treatment and to one that will not receive it. Here is where the ethical challenge comes in because many might argue that the decision they make might be influenced by preferences or biased based on an emotional standpoint.
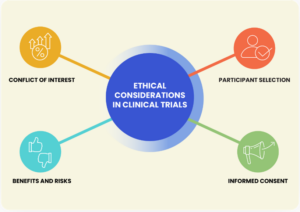
Clinical trials are also very selective, you must be within a certain category to be part of an experimental trial. The criterion of selection is based on the patient population that is being served by the practice, the type and phase of the trial, or the trial sponsor. Participating in a clinical trial can have a significant financial impact on someone. Thus, trial sponsors are those who cover the costs of the experimental treatment and any medical procedures.12However, there may be out-of-pocket costs for non-standard medical procedures or tests, in which these costs can add up, particularly for individuals with high-deductible health insurance plans. Some glioblastoma patients may not even have a close location to access a clinical trial. If they do, not all have the opportunity to participate. All these factors raise questions about equity and whether access to potentially life-saving treatments should be more widely available. No one should be rejected to participate only because they cannot afford it.
On the other hand, one of the biggest factors that play a role in the ethical decision-making of a patient’s health care is their family or legal guardian. The reason behind this is that their views can be different than those of the patients themselves. Some family members may advocate for more aggressive treatment options, even if the patient may have another preference. Balancing the perspectives and interests of patients and their loved ones can be tough for doctors to the point where legal matters may have to be taken.
Therefore, in order to avoid any conflict and issues that can arise as a patient’s health deteriorates, there are some premeasurements that can be made. For instance, when a patient is first diagnosed with an illness, whether terminal or not, the patient should be fully informed of their options and outcomes. Furthermore, they must require having a written voluntary consent stating the course they would like to take. If in this statement they provided information and are interested in wanting to participate in a clinical trial, then there are many legal matters that come as well. It is important to clearly explain the nature of the study, as well as any potential risks and benefits. They should also be aware from the beginning that they have the right to withdraw from these trials at any time without any financial consequences. Another key thing is to be transparent about the study’s objectives, and methodology, as well as any potential conflicts of interest.
All these principles and guidelines are essential for researchers to conduct a successful and ethical clinical trial. As well as ensuring the protection of participant rights and well-being. This not only maintains the trust of participants and the public but also advances medical science responsibly.
It is important to remember that at the end of the day, we are all just human beings who have the right to make decisions about our own health and what course of treatment we would like to take. As well as always being well-informed and transparent about all the options that can be taken. Glioblastomas, as we know, is one of the highest grades of brain cancer. They are extremely aggressive, and the tumors grow very rapidly. Due to this cancer being associated with a poor prognosis, it is challenging to treat even after being exposed to medication, chemotherapy and radiation. The next best treatment would be clinical trials. Although these trials may bring controversy and mixed feelings, sometimes they can be extremely helpful. They have allowed researchers to develop and evaluate new treatments and ensured that patients receive safe and effective care. Many clinical trials help save lives, enhance the quality of healthcare, and drive innovation in medicine. However, at the end of the day, it should all be up to the person to decide their own treatment.
- Brain tumors and brain cancer. Johns Hopkins Medicine. (n.d.). https://www.hopkinsmedicine.org/health/conditions-and-diseases/brain-tumor ↵
- U.S. Department of Health and Human Services. (n.d.). Genetics. National Institute of General Medical Sciences. https://www.nigms.nih.gov/education/fact-sheets/Pages/genetics.aspx#:~:text=Genetics%20is%20the%20scientific%20study,that%20help%20the%20body%20work ↵
- A brief guide to genomics. Genome.gov. (n.d.). https://www.genome.gov/about-genomics/fact-sheets/A-Brief-Guide-to-Genomics ↵
- The genetics of cancer. National Cancer Institute. (n.d.). https://www.cancer.gov/about-cancer/causes-prevention/genetics#:~:text=Other%20DNA%20changes%2C%20known%20as,is%20packed%20into%20the%20nucleus ↵
- YouTube. (2023). Inside Glioblastoma Multiforme: A Journey into the Deadliest Brain Tumor. YouTube. Retrieved October 22, 2023, from https://www.youtube.com/watch?v=WfBgAEpOIgs ↵
- Glioblastoma causes & risk factors. moffitt. (n.d.).https://www.moffitt.org/cancers/glioblastoma/diagnosis/causes/#:~:text=Like%20all%20cancers%2C%20glioblastoma%20is,types%20of%20astrocytoma%20brain%20cancer ↵
- Wei, D. C., & Morrison, E. H. (2023a, May 1). Histology, astrocytes – statpearls – NCBI bookshelf. National Library of Medicine. https://www.ncbi.nlm.nih.gov/books/NBK545142/ ↵
- MD Anderson Cancer Center, & Rose, L. (2022, February 22). Q&A: Understanding glioblastoma (GBM). MD Anderson Cancer Center. https://www.mdanderson.org/cancerwise/understanding-glioblastoma.h00-158828856.html#:~:text=Although%20glioblastoma%20can%20happen%20anywhere,brain%20stem%20or%20spinal%20cord. ↵
- U.S. Department of Health and Human Services. (n.d.). What are clinical trials and studies? National Institute on Aging. https://www.nia.nih.gov/health/what-are-clinical-trials-and-studies#what ↵
- Clinical trials: Cancergrace. The Society of Thoracic Surgeons. (n.d.). https://cancergrace.org/find-clinical-trials ↵
- Pace, A., Koekkoek, J. A. F., van den Bent, M. J., Bulbeck, H. J., Fleming, J., Grant, R., Golla, H., Henriksson, R., Kerrigan, S., Marosi, C., Oberg, I., Oberndorfer, S., Oliver, K., Pasman, H. R. W., Le Rhun, E., Rooney, A. G., Rudà, R., Veronese, S., Walbert, T., … Dirven, L. (2020, July 16). Determining medical decision-making capacity in brain tumor patients: Why and how?. Neuro-oncology practice. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7716185/#:~:text=Brain%20tumor%20patients%20are%20at,treatments%20or%20participation%20in%20research ↵
- Henderson, A. R. (2019, January 24). Informed consent: A process designed for you. Lilly Trials Blog. https://trials.lilly.com/en-US/blog/informed-consent-process-designed-you ↵



1 comment
Esteban Serrano
I really like this article because it does bring up a valid point. However, learning about brain cancer in particular was interesting. I didn’t know the different lobes of the brain, depending on where the tumor is located, can cause different symptoms.
Thanks for the insightful article, Maria! Congrats on your nomination!