An anxious daughter brings her 62-year-old mother into the clinic for a neurological evaluation with concerns about her memory. She claims that within the last few years, she has noticed her mother forgetting important things, frequently asking for the repetition of information, and losing track of time easily. Her inability to grasp and retain new information, along with her changes in mood, had caused her to experience a social withdrawal, which became drastically more significant after the death of her husband. She became increasingly concerned once her mother demonstrated difficulty with verbal communication. She was still able to perform her usual tasks, such as dressing herself, preparing her food, and cleaning her house, but performing a new task was strenuous. The daughter noticed that it became difficult for her mother to handwrite; she was slower, and her handwriting was less legible. After the initial consultation with the patient and her daughter, the practitioner first observed her medical history, in which it was uncovered that the mother had type 2 diabetes. They were also able to discover that the patient’s father suffered from dementia. The physical examination of the patient included neurological exams such as cognitive assessments as well as brain imaging. The patient demonstrated a lack of muscle strength, difficulty performing physical tasks such as standing up and sitting down independently (lost balance easily), as well as a loss of hearing and a slight loss of sight. She was unable to state the correct present date, month, or year. She was also unable to say what state she lived in. Registration and recall test demonstrated a lack of ability to retain information. When asked to state the names of objects she mostly referred to them as “things”, unable to name a clock or a necklace around her own neck. When asked to perform simple tasks such as folding a piece of paper and placing it on the desk, she became frustrated. MRI scans of the patient’s brain displayed shrinkage in certain regions of her brain, but blood tests, electrocardiograms, and cerebrospinal fluid tests revealed no abnormalities. After these examinations and the ruling out of several other brain disorders, the physicians diagnosed the patient with Alzheimer’s disease.1
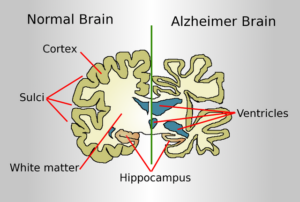
Alzheimer’s is an epidemic that is robbing more than 6 million people in America alone of enjoying the last few years of their lives comfortably. It is a degenerative disease in which the blood barrier of the brain is damaged and allows for the buildup of toxins such as amyloid beta and tau proteins. The nerve cells of the brain function like many other cells of the body; they require nutrients, they communicate with each other, they perform specialized functions, and they produce waste as a result of all of this. Brain cells are high-energy consuming cells, and as a result, they produce significant amounts of metabolic byproducts and interstitial waste products such as carbon dioxide, amyloid beta, tau protein, and lactate. A healthy brain is able to maintain homeostasis by the rapid exit of these toxins by waste clearance mechanisms. When normal functions of the cell, particularly waste clearance functions are altered, the accumulation of these toxins causes damage to the brain tissue.2
This results in the irreversible changes that cause the symptoms exhibited such as memory loss and unstable mood. In patients with Alzheimer’s, we observe a buildup of plaques and tangles. Protein buildups are a normal process that occurs with age, but this disease is characterized by an aggressive degeneration of the brain along with heightened buildup. Plaques are deposits of beta-amyloid, a protein fragment that is a metabolic waste product found in the brain’s interstitial fluid. This builds up between the nerve cells, preventing communication and signaling.3 We can also observe tangles; twisted fibers of the tau protein, which is meant to help stabilize the structure of the brain. In patients with Alzheimer’s, it clings to other proteins to cause these tangles.4 The medical community has not uncovered where this damage begins, but the greatest risk factors include age and family history. Late-onset Alzheimer’s symptoms first appear around the age of sixty, with early onset presenting as young as 30 years old.5 There is currently investigation towards the hypothesis that physical activity, diet, alcohol consumption, and smoking may be correlated.6
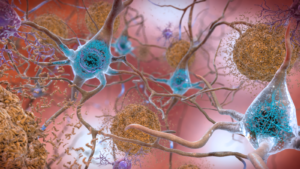
Our patient’s state began to decline more vigorously. She became increasingly unable to recall important information such as where she was and what day it was. She began to wander easily and get lost, she experienced difficulty in performing her usual tasks such as reading the paper every morning. She sometimes displayed a refusal to engage in these tasks such as brushing her teeth. Her family noticed increased hostility and sometimes delusions and hallucinations. She developed a habit of constantly wringing her hands. The family acknowledged that it was no longer suitable for her to live on her own, and she required constant care.
This moderate stage of Alzheimer’s is when the buildup of toxins and proteins in the brain causes inflammation and damage that makes it difficult for the patient to communicate effectively and perform routine tasks on their own.7 Damage to neuronal cells prevents them from performing their usual tasks and functions. The hippocampus is the area of the brain that plays a major function in learning and memory. In the hippocampus of a patient with Alzheimer’s, vacuoles that are filled with small particles cause granulovacuolar degeneration.8
The family became educated about the disease and were able to receive therapeutic services to aid in the process of understanding that the only route of treatment available is a medication that slows the progression of the disease and makes life a little bit more comfortable for the patient. The physicians introduce them to a clinical study that is testing a drug in development. However, after the conclusion of the trial, our patient’s state continued to worsen. The daughter was responsible for taking care of her mother, and she was eventually required to quit her job in order to provide around-the-clock care for her mother. It was a truly heartbreaking journey for the family to witness such a drastic decline in the mother’s state. Her physicians recommended that the patient engage in activities that could help maintain the health of the mind, such as sensory stimulation tasks and movement tasks like memory games and light exercises such as aerobics.
Clinical studies of non-pharmacological interventions, such as cognitive stimulation therapy, have been shown to improve the quality of life for patients.9 The brain is like a muscle, you must engage in activities that stimulate it in order for it to remain healthy. However, this is clearly not a route of curative treatment. Alzheimer’s disease was first recorded in the early 1900s by Dr. Alois Alzheimer, but it wasn’t until 1987 that the first Alzheimer’s drug trial took place (by Pfizer). A nationwide network was established by the National Institute on Aging that established an infrastructure for Alzheimer’s research.10 We have also identified a gene that is related to a form of Alzheimer’s disease that is inherited. Tacrine was the first drug approved by the FDA that treat memory loss symptoms, and it functions by reducing the breakdown of acetylcholine, a chemical messenger that helps the brain function properly. Major advances have been made in the monitoring and detection of the disease. Emphasis on the investigation of biomarkers has also developed. Patients diagnosed with Alzheimer’s are typically prescribed medications that help slow down the biological progression of the disease or lessen symptoms such as memory loss. Earlier this year, the FDA approved the use of Leqembi to treat Alzheimer’s patients; this drug reduces the number of amyloid plaques that were previously discussed by binding to the plaque and breaking them apart.11 This is a significant landmark in the journey of Alzheimer’s research because it guides us to the right direction as to what biological mechanisms must be targeted for improvement.

Our patient was given drugs that treated her agitation and mood swings. She began to lose control of her body at times and had several accidents in which she dropped objects easily and began to fall while doing normal daily tasks. One day she fell to her knees, and this resulted in a patellar fracture requiring surgery. During her recovery, she fell ill with what seemed to be a common cold and her health would simply not improve. The patient unfortunately passed away soon after.
Alzheimer’s is a devastating disease that places such a profound impact on the families of those affected. It currently affects more than six million Americans, and this number is expected to more than double by the year 2050, and an estimated 1.2 million more care workers will be required to care for patients with Alzheimer’s in the next ten years alone.12 The population of people who are suffering from Alzheimer’s are the people that once devoted their own lives to caring for us. We need not forget about the members of our community that have paved the way for our own success; I could think of no greater way there is to use the resources they have allowed us to seek out than to engage in more research and develop our treatment in order for the wise souls of our country to have a peaceful and gracious exit from this life. The subject of death is a very uncomfortable one, but when in the face of inevitable loss, we must not allow each other to have to walk this path alone and in distress.
While Alzheimer’s is a glooming gray cloud that grows larger and more daunting each year, there is plenty of hope and a large amount of advocacy to emphasize this area of research. In 2020, there was a hearing discussing the Alzheimer’s crisis regarding treatment pipelines and fiscal implications to the United States Senate including the Committee on Finance and the Subcommittee on Health Care. This hearing discusses the rising concern for Alzheimer’s research and the development of a cure and treatment. The National Alzheimer’s Project Act was signed into law in 2011 with hopes of finding a cure by the year 2025. We are approaching this deadline very quickly, and this hearing emphasizes the importance of pushing forward. Senator Toomey, the Chairman of the Subcommittee on Health Care, addressed an inspiring point about COVID-19. He states that the race to end this pandemic showed Americans the ability of our nation to develop and advance treatments when its people are in need, particularly regarding the pharmaceutical and diagnosis fields of medicine. However, he emphasizes that it is the alignment of incentives and public policy that really propels such incredible innovations to be made in the scientific community with regard to caring for our country’s devastating threats to public health.13 With our current understanding of the projections of Alzheimer’s disease cases in the future, I believe it is extremely important for us to create more incentives for the research of this disease. While we must be patient and understanding of our current capabilities and limitations within the scientific community, it is truly motivating to see how the public has advocated for Alzheimer’s awareness. The Alzheimer’s Association has established an Alzheimer’s and Brain Awareness month in June, along with setting purple as the official color of the movement. An important stand that has also been important is teaching the public that Medicare covers all FDA-approved drugs except for those that treat Alzheimer’s.14 This is a blaringly unfair disparity that makes absolutely no sense. This is a fatal disease, and when people do not have access to the healthcare and medication necessary to lessen the severity, it can make the disease much more intense for the patient and even those around them. Still, families all around the country are united in the fight to end the disease; the perseverance in this movement is a comforting reminder of how passion can drive even the most difficult of goals to be achieved. There is an annual Walk to End Alzheimer’s that gathers funds to be attributed towards care and support. People around the world are sharing their stories about caring for those afflicted with the disease. There is a lot of work that needs to be done in order to provide better care for the people who once nurtured us, but medical advancement is an unrelenting force that continuously develops miraculous growth and is fueled by our humanity and love for one another.
- Stages of Alzheimer’s. (n.d.). Alzheimer’s Disease and Dementia. Retrieved October 19, 2023, from https://alz.org/alzheimers-dementia/stages. ↵
- Kaur, J. et al. Waste Clearance in the Brain. Frontiers in Neuroanatomy 15, (2021). ↵
- Alzheimer’s Disease Facts and Figures. (n.d.). Alzheimer’s Disease and Dementia. Retrieved October 4, 2023, from https://www.alz.org/alzheimers-dementia/facts-figures ↵
- Tau Protein and Alzheimer’s Disease: What’s the Connection? BrightFocus Foundation. (n.d.). Retrieved October 20, 2023, from https://www.brightfocus.org/alzheimers/article/tau-protein-and-alzheimers-disease-whats-connection. ↵
- What Are the Signs of Alzheimer’s Disease? National Institute on Aging https://www.nia.nih.gov/health/what-are-signs-alzheimers-disease. ↵
- What is Alzheimer’s Disease? | CDC. (2023, January 7). https://www.cdc.gov/aging/aginginfo/alzheimers.htm ↵
- Stages of Alzheimer’s. (n.d.). Alzheimer’s Disease and Dementia. Retrieved October 19, 2023, from https://alz.org/alzheimers-dementia/stages. ↵
- Kurdi, M., Chin, E., & Ang, L. C. (2016). Granulovacuolar Degeneration in Hippocampus of Neurodegenerative Diseases: Quantitative Study. Journal of Neurodegenerative Diseases, 2016, 6163186. https://doi.org/10.1155/2016/6163186. ↵
- Behfar, Q., Richter, N., Kural, M., Clemens, A., Behfar, S. K., Folkerts, A.-K., Fassbender, R., Kalbe, E., Fink, G. R., & Onur, O. A. (2023). Improved connectivity and cognition due to cognitive stimulation in Alzheimer’s disease. Frontiers in Aging Neuroscience, 15. https://www.frontiersin.org/articles/10.3389/fnagi.2023.1140975. ↵
- Milestones. (n.d.). Alzheimer’s Disease and Dementia. Retrieved October 20, 2023, from https://alz.org/alzheimers-dementia/research_progress/milestones. ↵
- Commissioner, O. of the. (2023, July 7). FDA Converts Novel Alzheimer’s Disease Treatment to Traditional Approval. FDA; FDA. https://www.fda.gov/news-events/press-announcements/fda-converts-novel-alzheimers-disease-treatment-traditional-approval. ↵
- Alzheimer’s Disease Facts and Figures. (n.d.). Alzheimer’s Disease and Dementia. Retrieved October 4, 2023, from https://www.alz.org/alzheimers-dementia/facts-figures. ↵
- Grassley, C., Crapo, M., Roberts, P., Enzi, M. B., Cornyn, J., Thune, J., Burr, R., Carolina, N., Portman, R., Toomey, P. J., Scott, T., Carolina, S., Cassidy, B., Lankford, J., Daines, S., Young, T., Sasse, B., Wyden, R., Stabenow, D., … Masto, C. C. (n.d.). COMMITTEE ON FINANCE. ↵
- June is Alzheimer’s & Brain Awareness Month. (n.d.). Alzheimer’s Association | Alzheimer’s & Brain Awareness Month. Retrieved October 20, 2023, from https://www.alz.org/abam/overview.asp. ↵




3 comments
Dylan Vargas
Reading more about Alzheimer’s is a difficult task for most people. The disease attacks so many people all around the world and reading the article makes me understand how difficult Alzheimer’s really is, not just for understanding but for the people that are affected by the disease. Explaining things like the blood barrier of the brain is damaged and allows for the buildup of toxins such as amyloid beta and tau proteins, I always knew about Alzheimer but this information that I never knew about allowed me to grasp the basics of it. Of course, even with this well-done article, there is no real answer for the disease, and is always being studied.
Belkis Perrusquia
Alzheimer’s is a horrible disease, especially if a loved one is suffering through the disease or unfortunately carries the gene of the disease. It would be best to educate oneself about Alzheimer’s, especially if there is a history of this disease in your family and see what early actions they can take to delay it as long as possible.
Andrea Realyvasquez
It is extremely upsetting to know that more than 6 million Americans are affected by the influences of Alzheimer’s with family members being overcome with a feeling of helplessness. If your family has a history of the disease I believe it is so important to educate yourself and your loved ones about Alzheimer’s at an early stage in order to understand what measures can be put in place to impede its effects.